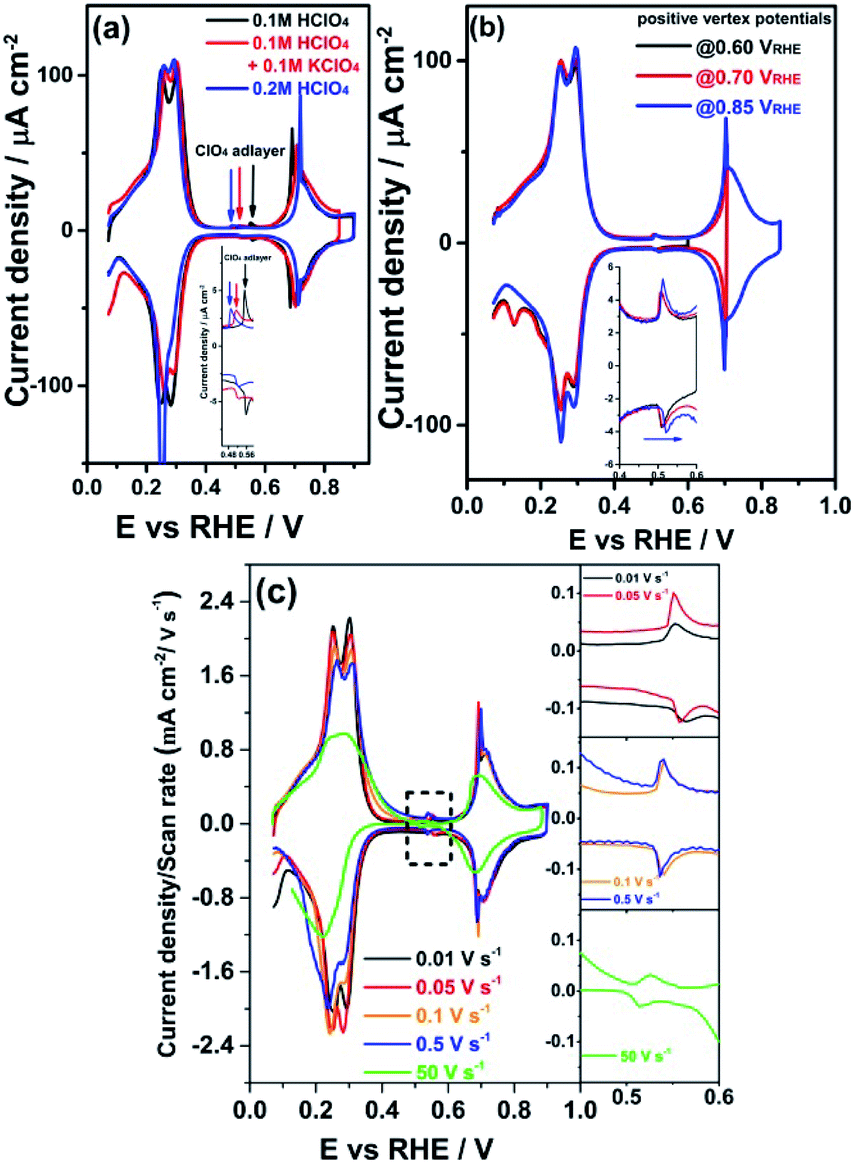
These ions present equal stoichiometric ratios. Explanation The oxidation number of an atom is the charge that atom would have if the compound was composed of ions.NO is a gas whose oxidation number is zero. The upper image represents the electrostatic interactions between the K+ cation and the OCl anion (with oxygen carrying a negative formal charge). Within a group (family) of elements, atoms form ions of a certain charge. This can be verified by comparing the oxidation number of Cl in KCl (-1) with that of Cl in KClO (+1). The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. It’s the holiday season, so you will be forgiven if you don’t think about all of this chemistry when you light your Christmas, Hanukkah, or Kwanzaa candles.\): Predicting Ionic Charges. The oxidation number of an element in a chemical compound is the charge it would have if it were the only element present in the compound. The O atom gains two electrons and becomes a +2 charge. Upon striking, the phosphorus ignites, liberating oxygen from the match’s KClO 3, which in turn ignites combustible substances (e.g., sulfur) in the matchhead. frequency of Cl 35 in KClO 3 at 273.16 °K and atmospheric pressure is 28,213,372 ☒ Hz. In the second reaction, the K atom loses one electron and becomes a +1 charge, while the Cl atom gains one electron and becomes a -1 charge. Modern safety matches contain no phosphorus but red phosphorus is embedded in the rough surfaces of matchboxes. Eventually, the toxic white phosphorus was superseded by the red allotrope. Addformal charges (minimized) to the atoms, remembering that theformal charges. Later on, white phosphorus replaced antimony sulfide to make matches more reliable. Create an equation for each element (K, Cl, O, H) where each term represents the number of atoms of the element in each reactant or product. Potassium chlorate (KClO3) isused in oxygen generators aboard aircraft. (2) If the substance is an ion (either there. For example, the H2O molecule exists as a neutrally charged substance. +1 Rules to remember when trying to find out the oxidation state of an element: (1) The total charge of a stable compound is always equal to zero (meaning no charge). x+1+ (-2) 0 x +1 Thus, oxidation state of chlorine in potassium chlorate is +1. When formed into matches, the mixture sometimes (but not always) ignited when struck on sandpaper. For your question, KClO4, based on rule 3, the answer is +1. The oxidation number of chlorine in KClO is: A + 1 B + 3 C + 5 D + 7 Easy Solution Verified by Toppr Correct option is A) K+1Cl O2 oxidation state of alrali metals +1 oxygen -2 Let oxidation state of chlorine is x. The use of KClO 3 in matches dates back to 1826, when English chemist John Walker combined it with antimony(III) sulfide, gum, and starch. As a high school or college chemistry student, you may have used it to generate oxygen in the lab.īecause it is a strong oxidizer, KClO 3 must be kept from contacting organic matter reduced inorganic materials such as elemental sulfur, phosphorus and iodine and concentrated acids. 
Step 2 :- To calculate the time required to. Number of moles Mass of the substance Molar mass 10 122 moles 6 OH-+ Cl- ClO 3-+ 3 H 2 O + 6 e-Here, the total number o charge transfer is 6 e-It is conducted that by 6F charge 1 mole of KClO 3 is obtained. Compound states like (s) (aq) or (g) are not required. Step 1: To find the charge produce to make 10g KClO 3. How do we know that in the given chemical equations what is the oxidation state of chlorine as chlorine can have +7,+5,+1,+3 and -7 as it's oxidation state. For example, C6H5C2H5 + O2 C6H5OH + CO2 + H2O will not be balanced, but XC2H5 + O2 XOH + CO2 + H2O will. Calculate the mass of chlorine needed to produce 100g of KClO4. Replace immutable groups in compounds to avoid ambiguity. 
It is or has been a component of explosives, fireworks, safety matches, and disinfectants. Ionic charges are not yet supported and will be ignored. Potassium chlorate (KClO 3) is a strong oxidizing agent that has a wide variety of uses.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
